Clinical Toxicology Units
1.- Introduction
The last 50 years have seen a tremendous upsurge in the chemical and pharmaceutical
industries, which has undoubtedly proven to be not only of great benefit to mankind,
but also resulted in a greater danger to exposure to hazardous products, increasing the
chances of suffering from a toxic-borne pathology, whether acute or chronic, sporadic
or epidemic, mild or deadly. There is also a social aspect to consider, particularly
evident in our culture, namely the mass consumption of drugs of abuse, and all the risks
that implies.
There are many dangerous and sometimes easily accessible medicines (psychoactive,
antiarrhythmic) within reach of the public. The supply of low-cost addictive substances
(cocaine, amphetaminic derivatives) is never ending, our daily life takes place alongside
strong chemicals, whether in the home (caustic cleaning products), in industry
(solvents, metals) or in agriculture (pesticides). The environment is heavily polluted
with organic and inorganic waste. In short, everyone is the potential target of a toxic
agent, regardless of sex, age or profession.
2.- Epidemiology of poisonings
The aforementioned exposure mechanisms have inevitably resulted in both acute
(overdoses of drugs of abuse, suicidal attempts involving medicines, gas inhalation) and
chronic (solvents, heavy metals) poisonings, whether accidental (children), food-related,
occupational or criminal. Overdoses of alcohol and other drugs and the ingestion of
medicines constitute a veritable epidemic in our society and result in three to five
visits a day to the emergency rooms of our country's largest hospitals. Is Spain it is
estimated that some 120,000 acute poisonings occur requiring healthcare treatment,
3,000 of them sufficiently serious to require admission to an intensive care unit, with
over 1,000 fatalities.
Chronic poisonings are more difficult to evaluate and quantify. Their insidious
symptoms, non-specific and sometimes multiorganic, along with a frequent lack of
association with an external toxic agent hamper diagnosis, resulting in official figures,
mostly involving the workplace, that undoubtedly underestimate the scope of the
problem.
The wide range of potential toxic agents, and the fact that some of the most
dangerous appear infrequently, make it essential that diagnostic protocols and suitable
therapies be readily available. It is also necessary that analytical methods be developed
that are tailored to requirements and that effective toxicological information be made
available, to which end the Ministry of Justice's Toxicological Information Service is no
doubt a contributing agent.
But the fact that Toxicology, and in particular Clinical Toxicology, is not officially a
medical specialty in Spain has created a vacuum in public healthcare, both in and out of
hospitals, that has negative repercussions in treatment, education and research. It is
this gap that the Clinical Toxicology Units aim to fill
3.- Concept of the Clinical Toxicology Unit (CTU)
A CTU is the functional union of two or more hospital professionals who combine their
efforts for the generic purpose of improving the treatment of poison victims who go to
a hospital for acute or chronic exposures.
There is no limit to the size of the staff, and the inclusion of people from the following
areas would be desirable: Emergency Room, Intensive Care, Laboratory, Pharmacy,
Psychiatry, Internal Medicine and Pediatrics. Other medical or occupational risk
prevention specialists can also be included. The CTU should incorporate several types
of healthcare professionals (doctors, nurses, pharmacists, biologists and others) who in
their day-to-day activities attend to poison patients in one way or another.
The common absence of a toxicology staff in hospitals, as well as of a toxicology ward,
are precisely what confer a functional character to these CTUs, whose services are
detailed below
The coordinator of these units must be a certified leader in the field of clinical
toxicology and the Unit must be explicitly recognized by the hospital and, ideally, by
local or state political-healthcare authorities. The recognition of the CTU by the
hospital itself and by the services to which its constituents belong is an essential
condition, since we are talking about a Functional Unit whose members join it freely and
voluntarily, though with the approval of those Services which must be provided in order
to comply with the mission of the CTU.
4.- Services offered by a CTU
The services offered and performed by a CTU can vary and depend primarily on the
healthcare environment in which it is located (type of hospital) and on the availability
and number of its staff. It should include, at least, the following five aspects:
4.1.- Treatment
- Standardization of Emergency Room and Intensive Care Unit treatment for
acute poisonings.
- Availability of an antidote first aid kit with administration and dosage
guidelines. Table 1 shows the CALITOX-2006 proposal.
- Collaboration with attending physicians to standardize a clinical guide to
poisonings.
- External consultation to track acute poisonings as required and to evaluate
chronic exposures
4.2.- Epidemiological control
- Logging of acute poisonings treated in Emergency Room
- Logging of poisonings treated in ICU
- Poison control: Specific record of exposure to chemical products
- Drug control: Specific record of overdose from drugs of abuse
- Record of work-related chronic poison exposures (first aid treatment).
- All of this information should be made available to healthcare officials, along
with preventive recommendations.
4.3.- Toxicological analysis
- Availability of qualitative and quantitative analysis on an emergency basis
(24h/day). Table 2 shows the CALITOX-2006 proposal.
- Scheduled availability of qualitative and quantitative analysis.
4.4.- Teaching
CTUs have a moral obligation to conduct on-the-job training of personnel taking part in
the treatment of poisoning patients at their own center through regular ongoing
training courses.
CTUs should participate in university toxicology training (undergraduate and graduate)
and in non-university courses and in the publishing of practical, easily accessible
(electronic) manuals.
4.5.- Research
CTUs should promote research in the field of Clinical Toxicology by requesting grants
(fellowships), contributing to national and international conferences and submitting
articles for publication in indexed journals.
5.- Operation of CTUs
The staff of a CTU should meet at least once per quarter to set objectives and
establish a method for achieving them. All tasks accomplished, always within working
hours, require time that must be made available by the Services involved. Committees
may be formed for achieving specific objectives.
6.- Location, structural requirements and cost
The functional nature of CTUs means they do not require a specific location, though it
would be reasonable for the Service where the CTU manager works to double as a
meeting place and as its administrative and bureaucratic headquarters. This would also
imply the need for administrative personnel to devote part of their time to the CTU.
The availability of a computer for handling epidemiological data and a toxicology
information database (like POISINDEX) is a must, as are other small expenses whose
costs, as a whole, would not exceed 10,000 euros/year, since personnel costs are nil,
these being provided by the Services themselves.
7.- Current status of CTUs in Spain
In Spain there are six units whose characteristics match those of the model described
above: Hospital Clínic (Barcelona), Hospital Clínico Universitario (Zaragoza), Hospital Rio Hortega (Valladolid), Hospital General (Valencia), Hospital Son Dureta (Palma de Mallorca) and Hospital de Sant Pau (Barcelona), although each has its own particular characteristics..
8.- Outlook for CTUs
The Clinical Toxicology Section of the Spanish Toxicology Association proposes and
supports the creation of these CTUs throughout Spain, in that they are considered to
contribute to better treatment of poisoning patients, to a knowledge of the
epidemiological reality of our society and to establishing the bases for preventive care..
Santiago Nogué y Ana Ferrer
September 2009
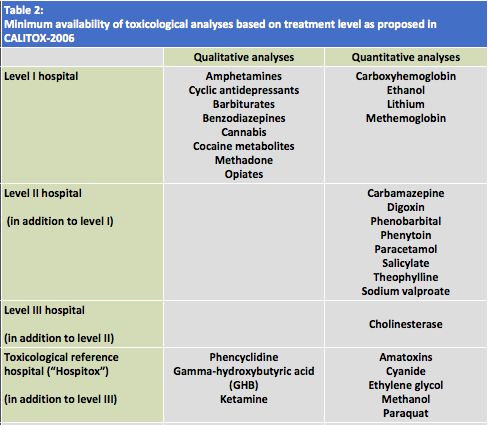
Table 1.- Minimum supply of antidotes and other medications of special interest in treating acute poisoning per CALITOX-2006 recommendations
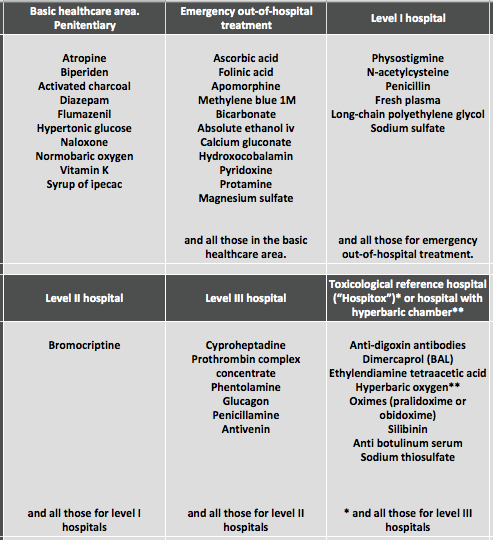
Table 2: Minimum availability of toxicological analyses based on treatment level as proposed in CALITOX-2006